OnCore
 OnCore® is a Clinical Trial Management System (CTMS) that has the ability to manage all components of a clinical trial in one central location. The software has the capacity to manage multi-site trials both nationally and internationally. The Oncore CTMS is managed through the WSU CRC; it is utilized university wide to support all aspects of clinical trials as the software has been adopted by faculty and staff in many Departments.
OnCore® is a Clinical Trial Management System (CTMS) that has the ability to manage all components of a clinical trial in one central location. The software has the capacity to manage multi-site trials both nationally and internationally. The Oncore CTMS is managed through the WSU CRC; it is utilized university wide to support all aspects of clinical trials as the software has been adopted by faculty and staff in many Departments.
Training and competency assessment are required to use the OnCore CTMS. Training may be scheduled on other dates upon request.
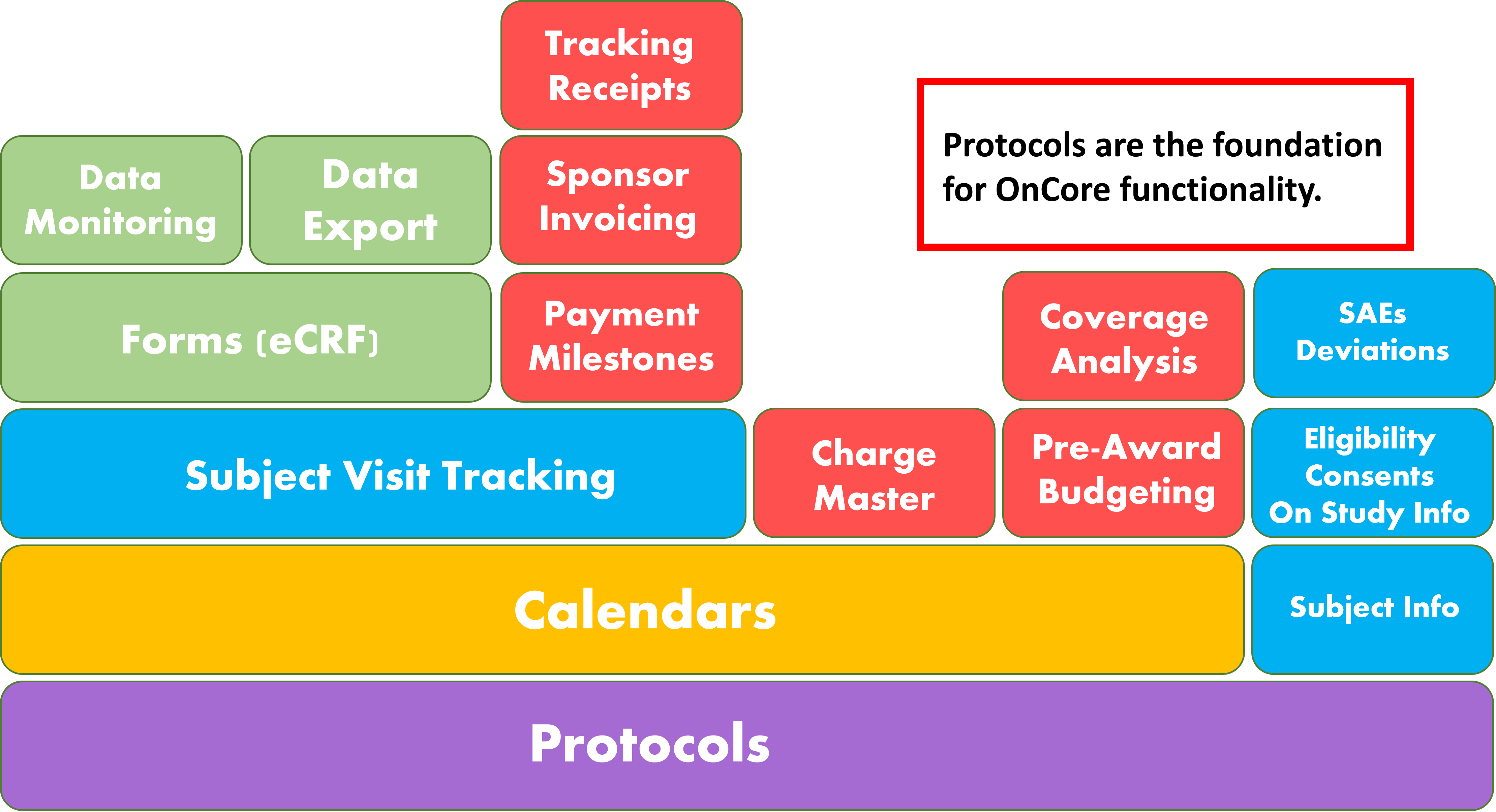
What Are the Benefits?
Increased Efficiency and Communication
Increased Patient Safety
System Integration
Multilevel Custom Reporting and Research Portfolio Review
Financial Management
Multicenter Study Management
Data Security
Data Management
Increased Regulatory Compliance
Register for OnCore Account